My video on the subject - 15/02/2026
Summary of AI Perplexity:
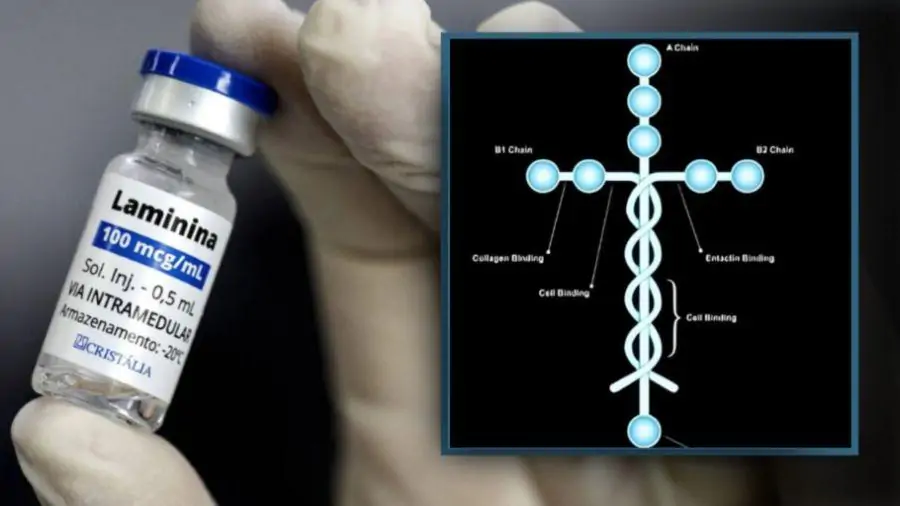
⚠️ Brazilian scientist Tatiana Coelho de Sampaio, a professor at UFRJ, has developed polylaminin, a protein extracted from the human placenta that regenerates damaged neurons in the spinal cord, allowing six quadriplegics to regain movement and walk after almost 30 years of research.
👇 Who is
Tatiana, a biologist, 59, UFRJ, heads a laboratory at the Institute of Biomedical Sciences. Mother of two and foster mother to a young orphan, she has dedicated 25-30 years to research, studied in several countries and avoids social networks to focus on “real life”, sleeping only 6 hours a night. Her team of 15 people has been shortlisted for the Nobel Prize in Medicine.
📚 What is Polylamine?
This experimental molecule acts as a “biological glue”, stimulating the reconnection of broken axons in the spinal cord, something considered irreversible by traditional medicine. Extracted from placental proteins, it is injected into the injury site to regenerate nerve circuits and restore mobility and sensitivity. Unlike palliative therapies, it attacks the neural cause, with human tests showing gains in the arms, legs and trunk.
🤜 Research began almost three decades ago, with initial tests showing partial recovery of movements. Investment of R$ 30 million is needed for scale production and final approval.
⚠️ Results
Six quadriplegics with serious injuries recovered voluntary movements: Luiz Fernando Mozer (motocross) felt sensitivity within 48 hours and contracted muscles; another 35-year-old moved his foot; Bruno Drummond de Freitas (31) returned to walking, dancing and driving a wheelchair; Diogo Barros Brollo moved his foot; Ruana Cruz (paralympic athlete) practiced adapted sports; and a 24-year-old from Cachoeira improved. Intensive post-application physiotherapy accelerated gains, restoring independence.
🤔 Current situation and future
The experimental treatment, pending regulatory phases by Anvisa for widespread use, is already rekindling global hope for millions paralyzed by accidents or violence.
🙏 And yes: it's true that the laminin (and its polymerized form, polylaminin) studied by Tatiana Sampaio has a molecular structure that resembles a cross.
--
On February 26, I consulted an AI tool to understand the current situation and discussions on the use of the scientific method. I asked the AI to answer the following question:
“Explain the controversy surrounding the compassionate use of polylamine and the questions about the application (or not) of the scientific method in validating its use.”
The answer:
In just a few months, polylamine has become a case study in how therapeutic hope, the media, legalization and science can collide. Today, the substance is promising and biologically plausible, but with clinical evidence is still in its infancy. The central controversy revolves around two things:
1) the widespread compassionate use via court decisions of something that is still in phase 1, and
2) whether the scientific method is being respected or trampled on - both in the research and in the way the product is communicated and used.
Below is an organized overview.
1. What is polylamine and what stage is it in?
- Polylaminin is a biomaterial derived from human placental laminin, an extracellular matrix protein involved in cell adhesion, axonal growth and neural plasticity.[1][2]
- The idea is to create, by polymerization, a “network” similar to that of embryonic laminin, capable of creating a microenvironment that favors axonal regeneration in the injured spinal cord.[1][2]
- There has been about 25-30 years of pre-clinical research on in vitro models, rodents and paraplegic dogs, with results considered consistent in terms of regeneration and functional recovery in animals.[2]
In humans, they have been published:
- An academic study of 8 patients with complete spinal cord injury, treated in the acute phase (up to 72 hours) with intramedullary application during surgery; according to the researchers, 75% had some motor recovery, compared to around 10% of spontaneous recovery described in the literature for this type of injury.[3][4][5]
- Additional case reports and small series, outside of randomized clinical trials, including compassionate use or court decisions, with improved sensitivity and/or movement in some individuals.[6][3][7]
From a regulatory point of view:
- On January 5, 2026, Anvisa authorized a phase 1 study to evaluate only the safety of intramedullary application in 5 patients with acute complete spinal cord injury (T2-T10, ≤72 h, with surgical indication).[1][8][9][10][11]
- Anvisa itself emphasizes that, at the moment, the study is insufficient to evaluate efficacy, and that if the safety data is favorable, only then can phases 2 and 3 come along to prove clinical benefit.[1][9]
In other words: scientifically, polylaminin is at the beginning of clinical development in humans (phase 1), even though there were previous experimental human cases and pilot series.
2. What is “compassionate use” in the case of polylamine?
Anvisa's Compassionate Use Program (PUC) allows new, promising and as yet unregistered drugs to be used in serious and debilitating diseases without satisfactory therapeutic alternatives, within specific regulatory criteria (technical dossier, responsibility of the company, doctor in charge, informed consent, monitoring of adverse events, etc.).[12][13][9]
In the case of polylamine:
- There is a regulated compassionate use program, linked to the Cristália laboratory, in which the drug is supplied free of charge, with application by trained staff and strictly controlled doses.[14][15]
- At the same time, there is a wave of judicialization: federal judges have granted injunctions ordering the inclusion of specific patients in compassionate use and the rapid release of the substance, often on the grounds that:
- the condition is extremely serious (recent tetraplegia/paraplegia, complete injury),
- there is no effective therapeutic alternative,
- there is a very short therapeutic window (ideally up to 72 hours, or up to 3 days, for greater response potential before spinal cord healing).[12][16][17][3]
- Court decisions in São Paulo, Minas Gerais and other states show this reasoning: they recognize the experimental nature, demand free and informed consent, but invoke the danger of irreversible damage and urgency as justification to intervene.[12][17][13][18]
By February 2026, news reports indicate:
- Dozens of legal applications (≥57 applications), with at least 24-28 patients having already received polylamine outside of a formal clinical trial.[5][15]
This use outside of a structured protocol is the main cause of the controversy.
3. How the controversy exploded: media, success stories and deaths
The case gained huge repercussions, partly because:
- Major media outlets have published reports talking about “reconnecting the spinal cord” and “restoring movement” to paraplegics/tetraplegics, with testimonials from patients who have been able to move their legs or arms again after an application.[3][19][2]
- Television and internet reports highlight the figure of 75% of patients with some motor recovery in the initial academic study (6 out of 8), compared to around 10% of spontaneous improvement expected in the literature, giving it a “historic breakthrough” tone.[3][4]
- The topic went viral: the leading researcher accumulated millions of interactions on social networks in just a few days, and personal reports of improvement were treated by the public as “proof of efficacy”.[20]
At the same time:
- Four patients who received polylaminine died recently, all as a result of use via court orders; according to the team, there is so far no evidence of a causal link with the substance, but the fact has intensified criticism and calls for strict monitoring of adverse events.[3][21]
- News reports indicate that, in the last 15 days, three of these deaths have occurred in patients treated by court order, reinforcing the concern of scientific organizations about the use of protocols that are not robustly monitored.[21][22]
This scenario (cases of improvement publicized as “miracles”, deaths with little transparency, avalanche of legal claims) is the backdrop to the discussion on compassionate use versus the scientific method.
4. Central criticisms: is the scientific method being respected?
4.1 Level of evidence available
Experts in evidence-based medicine have pointed out that:
- Data on humans is still limited to small, non-randomized series with no control group and case reports.[6][3][23]
- The 8-patient study itself, which supports the 75% improvement figure, had no control group, was not randomized and, in part, was not even published in a peer-reviewed journal when it began to be widely cited in the media.[24][25][23]
- There is a strong risk of:
- selection bias (who was included in the initial studies and who is able to sue);
- publication bias (positive cases are widely publicized, neutral/negative ones hardly appear);
- confusion due to co-interventions (surgery, early intensive rehabilitation, variability in the type and level of injury, etc.).[26][23]
Independent critical analyses (such as that of Prof. Dr. Wellington Lunz, UFES) classify the current level of evidence as very low, insufficient to speak of a “cure for paraplegics/tetraplegics”, even acknowledging biological plausibility and encouraging results in animal models.[23][27]
4.2 Judicialization and the breakdown of the research design
Scientific entities and bioethicists argue that the judicialized use of polylaminine..:
- It removes potential patients from controlled clinical trials, delaying the generation of robust evidence and paradoxically delaying broad and safe access to therapy.[26][28][21]
- Monitoring is fragmented: court-ordered applications are often not formally linked to the research protocol, and there is not always an obligation to systematically report adverse events or results to the scientific team.[29][21]
- It forces researchers to follow up scattered cases, without standardizing the time of application, inclusion and rehabilitation criteria, which “disorganizes” the research and compromises methodological rigor.[26][29]
SBPC and ABC editorials state that:
- Questions about scientific evidence should take place in technical forums, with peer review and methodological transparency, not via the media and case-by-case judicial decisions.[30][31][20]
- Isolated reports cannot be treated as “proof of efficacy”; the formal stages of clinical trials must be completed.[30][32]
4.3 Proposed follow-up without control group
The controversy increased when, in recent interviews (e.g. Roda Viva), the lead researcher considered carrying out later phases of the study without a control group, alleging ethical difficulties in denying the substance to patients and a possible “polylamine revolt”.[5][33][34]
- The scientific community reacted badly, because trials without a control group for a new and experimental drug fall outside the recommended methodological standard and drastically reduce the ability to infer causality (whether the improvement was due to polylamine, surgery, physiotherapy, the natural course, etc).[5][33]
- The researcher herself recognizes that the media repercussions “have gone beyond the usual pace of science” and that the excess of requests for compassionate use has hindered the regular progress of the research.[5][33]
For organizations such as SBPC and ABC, this type of methodological flexibility, motivated by social and media pressure, clashes with the core of the scientific method in clinical research: adequate comparison, control of bias, reproducibility and transparency of criteria.[30][37]
4.4 Scientific literacy and public trust
Another point of criticism is the potential damage to the scientific literacy of the population:
- An editorial in the journal Questão de Ciência compares the hype surrounding polylaminine to previous episodes (phosphoethanolamine, ivermectin, chloroquine, etc.), pointing out that although polylaminine is much more biologically plausible, the effect on scientific culture could be similar: early promise, inflated expectations, and possible frustration if subsequent clinical trials fail to confirm the “miracle”.[27][35]
- It should be remembered that, statistically, only around 15% of therapies that enter phase 1 actually prove to be safe and effective by the end of the clinical pipeline.[27]
- If the current narrative continues to be over-optimistic and, later on, the results are less impressive (or security problems arise), the risk is to reinforce the idea that “scientists don't know what they're talking about”, eroding public trust in science and institutions.[27][32]
5. The point of view of researchers and advocates of compassionate use
On the other hand, the research team itself and many advocates of compassionate use argue that:
- There are 30 years of pre-clinical data, with strong mechanistic plausibility and consistent results in animal models; this is not a “trendy vitamin”, but a biomaterial with a neurobiological basis.[2][25]
- In the initial human series, the rate of improvement (even partial) is much higher than that historically expected for complete spinal cord injury, which suggests a sign of efficacy that should not be ignored.[3][4]
- Typical patients seeking compassionate use are people with recent tetraplegia or paraplegia, with no proven effective alternative, a very high impact on quality of life and a high risk of irreversible damage. In these contexts, they consider that the ethical risk/benefit balance may justify offering an experimental treatment, as long as it is transparent and with informed consent.[12][16][13][18]
- Tatiana Sampaio herself recognizes that use outside of clinical trials is “scientifically incorrect”, but points out the human component: relatives and patients with no other options press for access, and judges end up deciding on a case-by-case basis.[24][25]
The Cristália laboratory, in a statement, emphasizes that:
- The supply for compassionate use is free of charge, the doses are strictly controlled and any commercialization of the substance is illegal, in an attempt to avoid ethical/commercial deviation from the process. [14][15]
The criticism of those who support this use is that regulatory bureaucracy and the slow pace of trials could deny a potentially unique chance to patients in a borderline situation; in this reasoning, primum non nocere is combined with the need not to “abandon” patients to the inexorability of tetraplegia while a plausible intervention exists.
6. Position of scientific societies and councils
Several organizations have recently spoken out:
- SBPC and ABC: joint letters and editorials calling for caution, technical rigour and a clear distinction between experimental research and consolidated treatment; they criticize the use of isolated reports as “proof” and warn of the risk of judicialization disrupting clinical development. [30][31][20]
- Texts from these organizations insist that methodological and evidence controversies should be dealt with in scientific forums, with peer review, research governance and data transparency, and not by pressure from public opinion and atomized judicial decisions. [30][32]
- COFFITO published a note reinforcing the strictly experimental nature of polylamine, without health registration, and pointing out that any use outside of approved clinical research is not supported by consolidated evidence or regulatory authorization for routine treatment.[36]
- The Senate announced a public hearing to discuss the use of polylamine, inviting Anvisa, the CFM, the National Academy of Medicine and other bodies, precisely because the issue has gone beyond the strictly technical domain and entered the field of public policy and regulation.[37]
At the time of writing, there has not yet been a formal, detailed opinion from the CFM with specific guidelines for the substance, but the tendency is for professional councils to take a stand as the debate progresses. [37]
7. To what extent is the scientific method being followed (or not)?
Summarizing the scientific-methodological point:
- Where the method is being followed
- There is a fairly classic path: a long pre-clinical phase, animal data, submission of a dossier to Anvisa, phase 1 authorization focused on safety, monitoring by ethics committees and Good Clinical Practices.[1][2][9]
- From a regulatory point of view, the pipeline is, in theory, aligned with the standard scientific method for developing drugs and biomaterials.
- Where there are deviations or tensions with the scientific method
- Extensive use outside of formal trials (compassionate use + injunctions) without standardization and structured data monitoring - this generates noise and adds little to the evidence, as well as disrupting the planned trial. [26][29][21]
- Public communication with a tone of “almost certainty that it will work” and “unquestionable technical result”, when the available evidence is, by EBM standards, still of a low level.[4][25][23]
- The proposal, although still under debate, of later phases without a control group, which goes against the usual methodological pattern for validating the efficacy of a new therapy.[5][33][34]
- Transformation of case series into “proof” in public perception, feeding disproportionate expectations and making it difficult to correctly understand that this is a promising hypothesis being tested, not a proven therapy. [30][27]
In strict terms, therefore, the scientific method is present in the regulatory design and intention of the trials, but is partially circumvented and distorted by the combination of:
- judicialization,
- media and social pressure,
- poor communication of the stage of evidence,
- and attempts to make study design more flexible in the name of humanitarian demands.
8. Critical summary
- Scientifically: polylamine is an experimental therapy with high biological plausibility and solid pre-clinical data, but still in phase 1 in humans, without controlled trials that allow efficacy to be affirmed with a degree of certainty compatible with evidence-based medicine. [1][6][9][23]
- Ethically: compassionate use, which is provided for in the law, can be justified in extreme cases with no therapeutic alternative, as long as there is informed consent, strict monitoring and coordination with formal research. The problem arises when this use becomes extensive, judicialized and poorly monitored.
- Methodologically: the biggest risk is not using polylaminin itself, but using too much, too soon and without proper design, which can:
- delay or misrepresent the generation of robust evidence;
- impair the ability to identify risks;
- and jeopardize public confidence if current promises are not confirmed.
In other words: the controversy is not whether polylaminine is “good” or not, but how to balance legitimate hope and scientific rigor. There is minimal consensus among serious organizations that the molecule is promising, that it deserves to be studied as a priority and that patients with spinal cord injuries desperately need new options. The divergence lies in how quickly and under what conditions to turn this promise into clinical practice - and to what extent the scientific method can be relaxed without eroding the very basis of trust that will ultimately allow us to say with certainty whether polylaminin works, for whom, how much, with what risks and in what windows.
My post on the subject - 26/02/2026
👉POLYLAMININE: when the hope of being able to move your arms and legs again comes up against fear, bureaucracy and scientific dogma.
The same “experts” who, during the pandemic, accepted still experimental products for billions of people are now raising all barriers against a substance that, in an extreme scenario and with no options, shows a consistent sign of benefit in acute spinal cord injury. Does this make technical, practical, ethical and human sense?
💪Some points:
- Decades of research and initial data in humans suggest a sign of efficacy, with a percentage of IMPROVEMENT WELL ABOVE that historically expected in complete spinal cord injury.
- There are still few patients and the therapy is still in the experimental phase. We need larger and better-designed trials, but that doesn't stop us from discussing compassionate use in “disillusioned” people, with a very short therapeutic window.
- Anvisa's Compassionate Use Program provides for the use of new and promising drugs in serious illnesses with no effective alternative, with a technical dossier, responsible doctor, informed consent and monitoring. In the case of polylamine, there is a structured program, free of charge, via the Cristália company, with trained staff and dose control.
- In any experimental treatment, there will be non-responders, adverse events and even deaths. If they are a minority, well researched and transparently reported, this does not invalidate the research or the judicious compassionate use.
😉 What about the “traditional” scientific method / Evidence-Based Medicine (EBM)? It's not a question of rejecting them, but of criticizing their rigid and selective use.
If we demanded double-blind trials for everything, not even a mother's love for her child would be “accepted” - there are no “randomized, placebo-controlled, double-blind studies or meta-analyses” of this. And no one would propose such a study by throwing people out of airplanes without parachutes to “prove” that parachutes work. There are situations in which biological plausibility, context and urgency are what should count the most.
--
The following text is my response to the “opinion request” you made on my social networks about the laminin controversy.
Do you mean that the same “scientists” who approved experimental products for use throughout humanity during the pandemic, without sufficient scientific studies at the time or clear and unequivocal proof that they worked and were safe, are now the ones questioning the safety and suitability of POLYLAMININE, which has already been preliminarily proven to give results for compassionate use?
What technical, practical or even ethical and human sense does this make?
Is it more important to be able to help people who may have already “lost hope” with some improvement, or to defend the “traditional scientific method” or crystallized “evidence-based medicine” at all costs?
Let's get to the FACTS, shall we?
- Polylaminin WORKS (the initial data strongly suggest a sign of efficacy, with a percentage of improvement well above that historically expected): the majority of patients treated with it, whether they were in the study or outside randomized clinical trials, including compassionate use or court decisions, obtained some motor recovery, with improved sensitivity and/or movement.
- There are still only a few studies and patients and it is still in the testing phase, but unequivocally (the preliminary data strongly suggests a benefit and makes it unlikely that it is all due to spontaneous improvement), it brings good results, which, statistically, would be very difficult to attribute to a placebo effect or “spontaneous improvement”.
- Anvisa's Compassionate Use Program (PUC) allows new, promising and as yet unregistered drugs to be used in serious and debilitating diseases without satisfactory therapeutic alternatives, within specific regulatory criteria (technical dossier, company responsibility, responsible doctor, informed consent, monitoring of adverse events, etc.). In the case of polylamine, there is a regulated compassionate use program, linked to the Cristália laboratory, in which the drug is supplied free of charge, with application by trained staff and strictly controlled doses. In other words, why try to hinder and criticize the access of patients who have already been “disillusioned” by conventional medicine to something that has real potential to help them, even if it is still in the experimental phase?
- Every treatment/substance that helps the majority will also always have some cases of failure and even poor results, and polylaminine is no different - being the exception and not the “rule”, this does not denigrate or “condemn” the therapy or the drug. To clarify: in any experimental treatment, it is expected that some patients will not respond or will have adverse events; cases of failure or death, if they are a minority and well evaluated, do not invalidate the investigation or the judicious compassionate use.
- We are in a time when some people absurdly argue that only quality or useful medicine that meets 100% the criteria of the rigid “traditional scientific method” or “evidence-based medicine (EBM)” is considerable, but much of what exists and has value simply cannot be judged from these perspectives; with due allegorical proportions and just to serve as an example, let's look at 2 “situations”:
When a mother says that she loves her child, the adherent of the “scientific method” and EBM should perhaps reject this premise and communicate it to the mother, since it is not possible to prove this subjective feeling through double-blind, randomized, placebo-controlled studies with a control group or meta-analyses, leaving only the “expert opinion” (the mother, in this case, evidence of the lowest relevance/reliability) on the issue;
The same supporter of EBM and the scientific method shouldn't trust parachutes as being effective either, since they can't be proven to work by double-blind randomized placebo-controlled studies or meta-analyses, leaving only “expert opinion” (in this case, those who have had to use them and survived, evidence of the lowest relevance/reliability) on the matter.
(In other words, do EBM and the scientific method need some flexibility, adaptations and improvements or, in fact, do they simply not “fit all”?)
Understand: it's not a question of rejecting the scientific method or EBM, but of recognizing their limits and preventing them from being used in a rigid and selective way, preventing plausible interventions in extreme contexts, such as acute spinal cord injury.
“Science is not an end in itself: its purpose is to help humanity” (Kátia Aguiar)
- It may be “scientific” to have a “control group” in every good scientific study, but in the case of the patients who benefit most from polylamine, would it be ethical, humane? Do you think it's appropriate that, in order to carry out a “good study”, part of the people who are already hopeless because they have serious health conditions (and who have been discouraged by conventional medicine) should be given substances that are known to be innocuous and have no effect? Or wouldn't it be more appropriate to try to “adapt the scientific method” when situations like this arise?
- When bureaucracy and excessive and inflexible technical rigor delay or hinder people's access to what could help them, doesn't this violate the Code of Medical Ethics, the Hippocratic Oath, good health practices and the priority of HUMANIZED care, in essence?
- Typical patients seeking compassionate use are people with recent tetraplegia or paraplegia, with no proven effective alternative, a very high impact on quality of life and a strong risk of irreversible damage. In these contexts, the ethical risk/benefit balance seems to strongly justify offering an experimental treatment:
Regulatory bureaucracy and the slow pace of trials could deny a “potentially unique” chance to patients in a borderline situation; in this reasoning, “primum non nocere” (one of the principles of medical activity) is combined with the need not to “abandon” patients to the inexorability of tetraplegia while a plausible intervention exists.
The excess of judicialization and supposedly technical questions/barriers, if not well articulated with the research protocols, can get in the way of producing the robust evidence that everyone wants, including the patients themselves.
- Some “scientists” seem to have been bothered by the fact that many people associate the cross shape of the laminin protein with religious issues (for example, that it could be a gift from God to humanity - if God created everything, it's worth remembering that the possibility cannot be logically ruled out...), to which researcher Dr. Tatiana Sampaio, in an interview, clearly clarified that if the shape of the protein is a FACT, people's interpretation of the fact is free, will be very individual and that she has no control or responsibility over this.
- Defending compassionate use here does not mean that the stage of controlled clinical trials is dispensable; on the contrary, they are essential if the therapy is to be offered safely and widely on the SUS.
By Dr. Ícaro Alves Alcantara
Quotes:
[1] Anvisa: Clinical research authorized to evaluate ... https://www.crfgo.org.br/noticia/id/1116/noticia-titulo/anvisa-pesquisa-clinica-e-autorizada-para-avaliar-a-seguranca-do-uso-da-polilaminina-em-humanos
[2] Polylaminin and the ways to regenerate bone marrow https://www.sciencearena.org/noticias/polilaminina-e-os-caminhos-para-regenerar-a-medula/
[3] Polylamine: Brazilian research reconnects the spinal cord of paraplegics https://g1.globo.com/fantastico/noticia/2026/02/22/polilaminina-a-substancia-que-reconecta-a-medula-e-traz-esperanca-para-pessoas-com-paraplegia.ghtml
[4] “The technical result is not open to question,” says Tatiana Sampaio about the polylamine study https://cultura.uol.com.br/noticias/74142_o-resultado-tecnico-nao-e-passivel-de-questionamento-diz-cientista-tatiana-sampaio.html
[5] Polylaminin: Tatiana Sampaio defends research data and considers study without control group https://jc.uol.com.br/colunas/saude-e-bem-estar/2026/02/24/polilaminina-tatiana-sampaio-defende-dados-da-pesquisa-e-cogita-estudo-sem-grupo-controle.html
[6] Polylamine: what is known so far about the treatment ... https://www.medway.com.br/conteudos/polilaminina-o-que-se-sabe-ate-agora-sobre-o-tratamento-experimental-para-lesao-medular/
[7] Brazilian experimental drug shows advances in ... https://www.portaltri.com.br/noticias/42553/medicamento-experimental-brasileiro-mostra-avancos-em-pacientes-paraplegicos
[8] Anvisa authorizes clinical research to evaluate safety ... https://site.cff.org.br/noticia/Noticias-gerais/06/01/2026/anvisa-autoriza-pesquisa-clinica-para-avaliar-a-seguranca-do-uso-da-polilaminina-em-humanos
[9] Anvisa authorizes clinical research to evaluate the safety of using ... https://www.gov.br/anvisa/pt-br/assuntos/noticias-anvisa/2026/anvisa-autoriza-pesquisa-clinica-para-avaliar-a-seguranca-do-uso-de-polilaminina-em-humanos
[10] Ministry of Health and Anvisa announce approval of study ... https://www.gov.br/saude/pt-br/assuntos/noticias/2026/janeiro/ministerio-da-saude-e-anvisa-anunciam-aprovacao-de-estudo-clinico-para-tratamento-inovador-de-lesoes-na-medula-espinhal
[11] ANVISA authorizes phase 1 clinical study with Polylaminine in Brazil https://bcrj.org.br/anvisa-autoriza-estudo-clinico-de-fase-1-com-polilaminina-no-brasil/
[12] TRF3: Federal Court authorizes compassionate use of polylamine ... https://www.sedep.com.br/noticias/trf3-justica-federal-autoriza-uso-compassivo-de-polilaminina-para-paciente-tratar-lesao-da-coluna-cervical/
[13] Federal court in Minas Gerais authorizes use of experimental drug ... https://sjmg.trf6.jus.br/justica-federal-mineira-autoriza-uso-de-medicamento-experimental-para-paciente-com-lesao-medular/
[14] Clarification note - Cristália https://www.laboratoriocristalia.cl/releases/nota-de-esclarecimento
[15] Patient in Goiás receives polylamine by court decision - via @podergoias https://www.podergoias.com.br/materia/25357/paciente-em-goias-recebe-polilaminina-por-decisao-judicial
[16] HUOP uses polylamine to help patients with ... https://www.parana.pr.gov.br/aen/Noticia/HUOP-aplica-polilaminina-para-assistencia-paciente-com-lesao-medular
[17] Federal Court authorizes compassionate use of polylaminine ... https://web.trf3.jus.br/noticias-sjsp/Noticiar/ExibirNoticia/2048-justica-federal-autoriza-uso-compassivo-de-polilaminina
[18] Polylaminin and Access to Health: A Legal Analysis of ... https://favarettoadv.com.br/polilaminina-e-acesso-a-saude-uma-analise-juridica-da-decisao-que-autorizou-uso-compassivo/
[19] Placental protein helps recover movement after injury ... https://www.faperj.br/?id=858.7.1
[20] Scientific societies call for caution and precision in debate on ... https://www.acessa.com/noticias/2026/02/311416-sociedades-cientificas-pedem-cautela-e-precisao-em-debate-sobre-polilaminina.html
[21] Researchers urge caution in polylamine debate https://www.poder360.com.br/poder-saude/pesquisadores-pedem-cautela-em-debates-sobre-polilaminina/
[22] Scientific organizations warn of risks in the use of ... https://folhadosudoeste.com.br/2026/02/21/entidades-cientificas-alertam-para-riscos-no-uso-da-polilaminina/
[23] Polylamine: NO Evidence of Cure for Paraplegics or ... https://www.wellingtonlunz.com.br/post/polilaminina-nao-cura-paraplegicos-ou-tetraplegicos
[24] Polylamine: the molecule discovered in Brazil ‘by chance’ ... https://www.bbc.com/portuguese/articles/c5y3zvv822jo
[25] ‘It's not a surprise, it's expected to work’, says researcher ... https://www.bbc.com/portuguese/articles/c0q3l8gk759o
[26] Pro-polylamine decisions could delay research and not ... https://www.acessa.com/noticias/2025/12/300679-decisoes-pro-polilaminina-podem-atrasar-pesquisas-e-nao-seguem-criterio-tecnico-afirmam-especialistas.html
[27] Polylamine and the damage to scientific literacy https://revistaquestaodeciencia.com.br/artigo/2026/02/20/polilaminina-e-o-dano-ao-letramento-cientifico
[28] Judicialization of polylamine use hinders research https://revistaoeste.com/saude/judicializacao-do-uso-da-polilaminina-dificulta-avanco-das-pesquisas/
[29] Judicial access to polylamine does not include clinical follow-up https://www.poder360.com.br/poder-saude/acesso-judicial-a-polilaminina-nao-inclui-acompanhamento-clinico/
[30] Scientific organizations call for caution over the use of polylamine https://gazetadoparana.com.br/artigo/sbpc-abc-cautela-polilaminina-lesao-medular
[31] Polylamine: scientific organizations urge caution to avoid ... https://portal.sbpcnet.org.br/noticias/polilaminina-entidades-cientificas-pedem-cautela-para-evitar-expectativas-desproporcionais-com-o-medicamento-ainda-em-testes/
[32] Scientists urge caution over polylamine, a molecule tested in the ... https://www.diariodenoticias.com.br/materia/cientistas-pedem-cautela-sobre-polilaminina-molecula-testada-no-tratamento-de-lesoes-medulares
[33] Tatiana Sampaio defends the study of polylaminine without a ... https://viva.com.br/saude-e-bem-estar/tatiana-sampaio-defende-fazer-estudo-de-polilaminina-sem-grupo-controle.html
[34] Polylaminin: Tatiana Sampaio defends research data and considers ... https://istoe.com.br/polilaminina-tatiana-sampaio-defende-dados-da-pesquisa-e-cogita-estudo-sem-grupo-controle
[35] Polylamine and the damage to scientific literacy https://www.revistaquestaodeciencia.com.br/artigo/2026/02/20/polilaminina-e-o-dano-ao-letramento-cientifico
[36] Federal Council of Physiotherapy and Occupational Therapy https://www.coffito.gov.br/nsite/?p=37287
[37] Public Hearing to discuss the use of Polylaminin in ... https://www12.senado.leg.br/radio/1/noticia/2026/02/25/audiencia-publica-vai-discutir-uso-da-polilaminina-em-lesoes-medulares
[38] General News https://site.cff.org.br/noticia/Noticias-gerais/03/02/2026/polilaminina-avanca-em-testes-clinicos-e-reacende-expectativas-no-tratamento-de-lesoes-medulares
[39] Information about Polylaminine https://www.cristalia.com.br/releases/informacoes-sobre-a-polilaminina
[40] Polylamine could be offered free of charge by ... - CFF - News https://site.cff.org.br/noticia/Noticias-gerais/25/02/2026/polilaminina-podera-ser-oferecida-gratuitamente-pelo-sus-apos-aprovacao-regulatoria
[41] Polylamine - Wikipedia, the free encyclopedia https://pt.wikipedia.org/wiki/Polilaminina
[42] Polylaminin: Tatiana Sampaio defends research data and considers ... https://circuitomt.com.br/polilaminina-tatiana-sampaio-defende-dados-da-pesquisa-e-cogita-estudo-sem-grupo-controle/
[43] A patient who received polylamine by court order reports ... https://www.cristalia.com.br/midia/paciente-que-recebeu-polilaminina-por-ordem-judicial-relata-emocao-ao-mexer-o-pe-e-a-perna
[44] Latinofarma - Cristália https://www.cristalia.com.br/latinofarma/en
[45] If a patient gets better after receiving a substance... that's ... https://www.instagram.com/reel/DVJTA_egNlJ/
[46] Anvisa authorizes clinical research to evaluate the safety ... https://rn.cff.org.br/noticia/Noticias-gerais/06/01/2026/anvisa-autoriza-pesquisa-clinica-para-avaliar-a-seguranca-do-uso-da-polilaminina-em-humanos
[47] Polylamine and the right to recovery - Migalhas https://www.migalhas.com.br/depeso/449523/polilaminina-e-o-direito-a-recuperacao